🎧 Here on The ChinaHealthPulse Podcast, I chat in depth with the real experts who have dedicated years to working in and with China’s health - across policy, industry, academia and well beyond. Our candid conversations aim to provide you with real insight into how care is delivered, how decisions are made, and why it all matters, far beyond China’s borders.
Watch or listen here on substack or Youtube, and/or subscribe on Spotify & Apple Podcasts. These newsletter posts provide all links, plus a full text transcript of each episode.
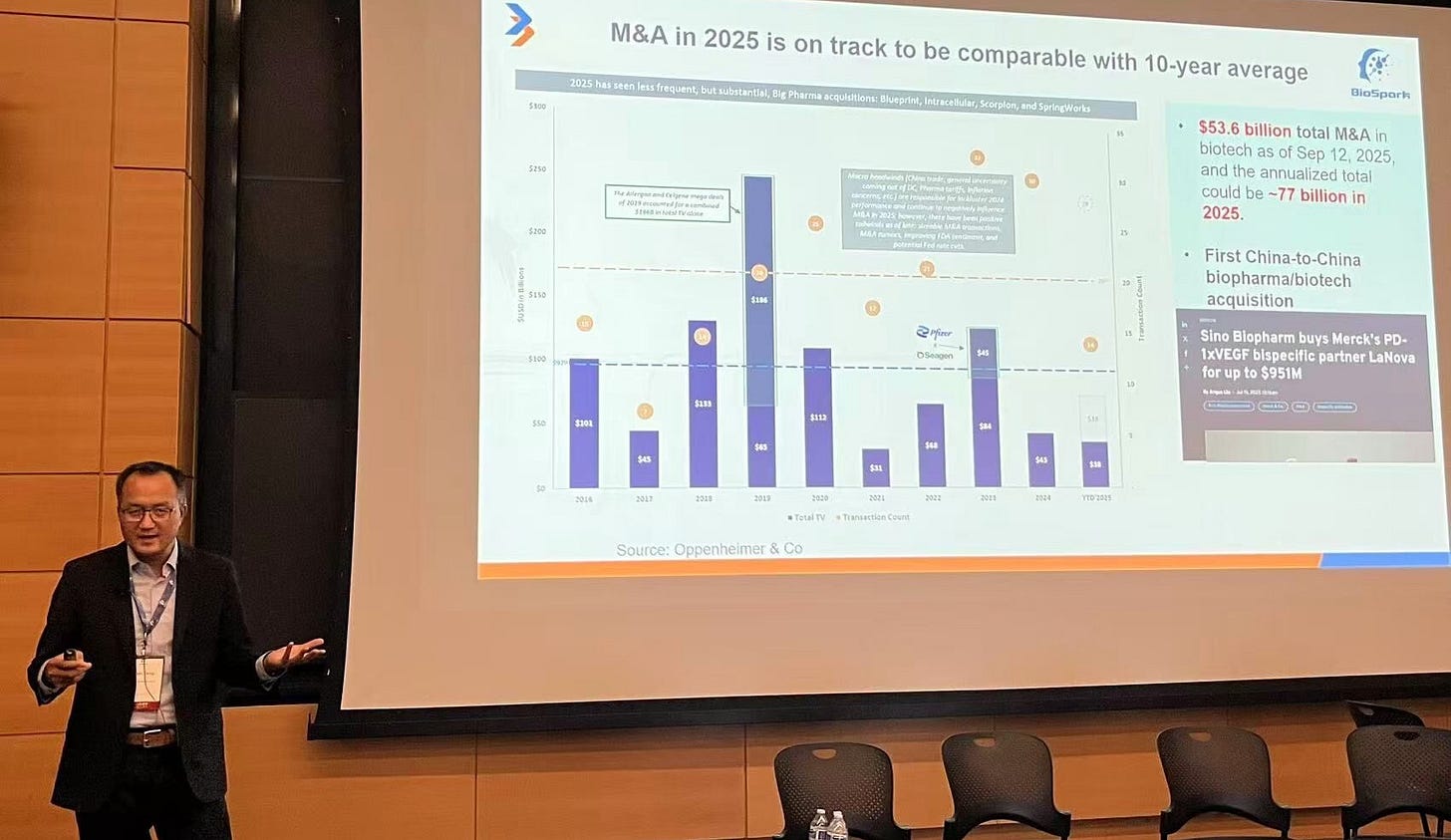
It’s March 2026: Q4 2025 is done, end-year numbers have come in, and annual reports shared through summary earnings calls over recent weeks have set the tone for how biotech capital will move in 2026. We’re also a few months out from JPM 2026, where there was even more China-West biotech deal chatter than last year, if possible.
Now is therefore the perfect moment to zoom out and translate the noise into a clearer map: what is emerging across deals and financings, why they’re emerging now, what a ‘global-grade’ China-origin asset really means in practice.
I can think of no one with a better read on the landscape from an investment point of view, than Dr Leon Tang, one of the leading China biotech experts around, who sits closest to the investor and partnering circuits and always has his finger on the pulse.
Leon brings almost two decades of experience in biomedical research, BD and investment. He is a founding partner of InScienceWeTrust BioAdvisory, a biotech business development consulting firm that focuses on pharmaceutical licensing between the East and the West, which has advised over 30 clients ranging from Fortune 500 companies, world-leading investment funds, leading public and private biotech companies, and stealth biotech startups. He is the BD head of Mianus Accelerator – a new venture that aims to help Western biotech companies to access China’s world-class first-in-human clinical development capability. He is also the co-founder of the InScienceWeTrust Community, an Asian-biotech focused non-profit, and one of the most active Asian biotech communities I know, with over 4000 active members across global hubs, including in North America and Europe.
In addition, he is scientific advisor at BioSpark Group, with past lives in the venture capital and academic worlds. He holds a PhD in Biomedical sciences from the Icahn School of Medicine at Mount Sinai, a Master’s at Nankai University in Biochemistry, and a Bachelor’s from Tianjin University in biochemical engineering, and has published over 50 academic papers, including in Nature Reviews Drug Discovery and the Lancet Oncology.
Watch/listen/read on Substack, on Youtube, or subscribe to the audio podcast on Spotify and Apple
Read our Conversation:
(Audio transcript adjusted for clarity and flow)
1. Setting the scene
Ruby: Let’s get started by setting the frame. From your vantage point across networks and portfolios, including investors, BD teams and the wider biotech community, what feels most different right now, whether that’s compared with six months ago, a year ago or even further back?
Leon: if you ask me what’s the biggest difference compared to what I saw six months ago, obviously I’m a little bit biased, always looking at a lot of stuff from the lens of Asian biotech, particularly China biotech. I would say that the overall industry, especially the Western side when they look at China, the sentiment is much more positive compared to six months ago.
And if you go a little bit further back, even to one year ago, there was still debate whether embracing China was a good idea. There was an executive order rumoured from the Trump administration to ban the license deal from China to the US and so on. So that conversation and chatter is now much, much quieter today than before.
In the meantime, I would that say since JP Morgan in January, there’s a lot of high profile deals and collaboration. And the headlines announced really embrace the China biotech as an essential part of the global R &D ecosystem - through the deal making, through R &D collaboration, even through high profile big pharma executive visits.
So I think that would be the biggest difference: the sentiment and also the action with China biotech is much more positive compared to six months ago and much, much better compared to one year ago.
Ruby: And in terms of the recent end-year summaries from industry across biopharma, especially for those with China cash flows or China pipelines or cross-border ambitions, what have been the real questions for you underneath those very optimistic announcements, whether that’s real capital or real partnerships; opportunities and risks, things like that.
Leon: In terms of cash flow, think there are very few China originated drugs probably except for Zanubrutinib (Brukinsa) from BeiGene/Beone, and Carvykti (Cilta-cel) from Legend Biotech, who are making profit for Western companies. But most of others were only recently approved, and have not really reported revenue from the Western companies’ earning calls yet.
I’m probably more focused on the deal making front, licensing deal or R &D collaborations. For dealmakers, there are always two parts. One part is really for your balance sheet or for your CapEx/upfront payment, and then the milestones, everything biobucks (contingent, milestone-based payments in biotechnology licensing or acquisition deals between large pharma and smaller biotech firms), that’s a different category, which is really important for the Chinese biotechs to talk to their investors, but not necessarily have an immediate impact with the earning calls every quarter.
So focusing upfront payments, think if you look at all the deals since the beginning of 2026, the momentum suggests that this year can be bigger than 2025, which was already record breaking year in upfront payments. Biobucks has the same story. Last year, the global BD&L (business development and licensing), the licensed pharmaceutical space, China to West deals contributed to about 49% - about half the total deal value. As a comparison, in 2024, the United States were contributing about 50%, I think 47%. So from 2024 to 2025, the leader in licensing deals changed from United States to China. If you just look in the first two months of 2026 number already, China going to be dominant. It’s pretty clear. it continues.
Related:
2. Deal trends: Part 1
Ruby: In the pharmaceutical licenses you just mentioned, the big headline grabbing China-West partnership announcements that we’ve seen over the previous years, and then the burst of attention around January in San Francisco at JPM. In terms of organising the landscape then, what have been key patterns you’ve noticed in assets? any surprises or revisions in terms of modality, whether that’s ADCs versus bi-specifics, or clinical versus pre-clinical stage or buyer type - large foreign MNCs versus funds or other biotechs or even in structures for co-development or M&A, what they’re telling you about demand and innovation progress or pace and talent, or perceptions versus reality?
Leon: This is probably gonna take two hours to answer to really give you a little bit of a reasonable description, so I have to force myself to three. The first one I would say is diversity. So the China-to-West deal right now is diversifying away from previously, which pretty much mainly focused on oncology licensing deals. Two years ago, that was pretty much the majority of them. But today is really different. Diversification in terms of disease area. Yes, oncology is likely still number one, probably not far ahead of immunology, if you look at recent deals in the first two months of 2026 . And also obesity is catching up real quick.
The format of deals are also diversifying quite a bit: if you go back three years ago, most of the deals would be straightforward license deals. Right now it has so many different flavours (except for straightforward large ticket M&A, which is still not happening very often). Everything else is very similar to what you would say. You have a typical license deal with a preclinical phase one, phase two, phase three assay.
Actually this week, Pfizer’s licensed a commercial-ready product from the China biotech Hangzhou Sciwind Biosciences,and I just saw the news that that asset was approved in China, shortly after the deal was closed. So right now Pfizer is a commercial obesity company. So they have a commercial product on the market as of today, because they licensed a late stage asset (Ecnoglutide, a GLP-1 drug) from China. But of course this asset was only approved for Chinese market. So that’s a new development: big Pharma licensed a late-stage asset only for China market! That’s one diversification.
Another is: there’s a deal which even with the US biotech companies, rarely happens. Eli Lilly had a deal with Innovent, which is arguably the number one biotech based in China now, by market cap, because BeOne, or what used to be called Beigene, has become a Swiss company. So Innovent has become number one. Innovent and Lilly’s deal: if you look at it, there’s no asset transaction, but upfront payments of $350 million with up to more than $8 billion biobucks - one of the largest deals in 2026.
And there were no asset transactions involved. What Eli Lilly really got from this $350 million down payment, is really to have the option to co-develop drugs from target nominations until phase two with Innovent, in oncology and immunology. So what Eli Lilly got is not a specific asset, but Innovent’s capability to develop drugs in immunology and oncology.
And a similar situation also happened in the largest deal on record as of today, between CSPC and AstraZeneca. That deal, the first half probably involves four assets in obesity, and the other half is the option to develop another four obesity assets they don’t even have yet. And that deal costs AstraZeneca over $1 billion upfront payment, up to $18.5 billion total value. It’s the largest deal out of China.
So those also showed the diversification, also the confidence and the trust of big pharma like Lilly, like AstraZeneca. In China, biotech has the capability to develop drug from early stage all the way until phase two. I think that’s the first one.
Related:
3. Changing perceptions
Ruby: We’re talking about not just partnering on assets, but partnering on capability, or even the last example you said, the potential for capability - not even capability realised. So all of these stages really showcasing a huge amount of trust and hope in what Chinese skillset and talent and capability can bring to the table. That’s such a huge transition in the recent years. It’s incredible.
Leon: Right. But if we think about this happening exactly to some of Japanese and American biotech, I’ll give you two historic references. The deal Sanofi did with Regeneron, many, many years ago, Regeneron was a tiny biotech, it didn’t have the capability to do late stage clinical development, not even to mention commercial capability. That’s why they had this extensive collaboration with Sanofi. Sanofi’s best-selling drug on their pipeline right now, was originated from Regeneron, Dupixent/dupilumab. So you see the Chinese are following very similar footsteps as what Regeneron did, to become today as one of the largest biotech player in the world.
Another example is the US’s Alnylam, which had a lot of collaborations. One of the best cholesterol controlling drugs (Inclisiran/Leqvio) originated from Alnylam, went to The Medicines Company, then got acquired by Novartis. That’s probably the most innovative commercial stage cholesterol controlling drug on the market.
Ruby: Do you think it is because the momentum has built to a level where there is this trust that wasn’t there before?
Leon: There’s trust, there’s also recognition of each other’ capability. It’s not like China never changed and all of a sudden the Western companies discovered them. It’s an iteration process. Let’s just use AstraZeneca, for example. I think they have around 14,000 employees in China, and I assume majority of them are ethnic Chinese, and were trained in China. But of course there must be a lot of their global talents that were trained or worked in different countries, a different culture background. A lot of them must come from Europe. The people are very fluid: they change jobs, so over time, they’re going to build trust.
Hengrui - they have a senior executive who recently joined. She was a senior executive at AstraZeneca. And Hengrui’s BD head was a former executive from the BD team at J&J. Hengrui’s BD team, most of them had worked in a Western big pharma biotech investment firm before. So they know how they’re connecting the trust, naturally exchanging on a personal level or professional level.
Before any massive core development collaboration deal is announced, hundreds of billions worth of asset transactions will have already happened. So the R&D team from, let’s say, Lilly and Innovent knew each other really well - actually Innovent received investment from Lilly many, many years ago. So this deal is their seventh or tenth deal. They start with a straightforward and simple - I sell you buy an asset, give you rights - that kind of simple integration, until right now it has become a full, almost marriage type of collaboration. It’s a gradual build up. So I see all of this as a very natural integration of big pharma with late stage clinical development capability and commercial capability, and China based biotech company that really proves themselves, who are cost effective and deliver high quality early stage research in a lot of disease areas. So I think it makes sense for both companies, and for both sides.
4. Deal Trends: Part 2
Ruby: You mentioned there were three main types of trends and you talked about diversification first.
Leon: Diversity application is one. The second one, I would say definitely the influx of Western investment fund. Obviously, there are a lot of geopolitical barriers. There were many Western investors in China, I would say five or 10 years ago, especially in biotech. And then a wave of them left China, and then there was like a “China Rehab” capital challenge. And now they’re coming back.
There’s an incoming conference in late March called CHIC, the China Healthcare Investment Conference in Shanghai. The majority of their speakers are Tier 1 Western investment funds, senior investors. The Western investment fund are coming back. There’s Deerfield, there’s RA capital, there’s OrbiMed, there’s TCG Crossover Capital, you name it. Most of them are Western investors, most of them come from the United States, despite the uncertain US-China relationship. Despite all the geopolitical issues and the regulation and the government roadblocks, investors, are very, very pragmatic and resourceful. They figure out some way, because China’s early stage R &D capability just has so much more advantage compared to the Western biotech infrastructure or ecosystem. The investors just cannot let it happen without their capital.
The third trend is really on the regulation front. China’s regulatory agency used to be called CFDA. That just tells you how much the Chinese regulators admired the (US) FDA! That’s the name, before they changed it into NMPA, right? People used to say that China is a copycat. But guess what, the Chinese NMPA and Chinese government regulators are now coming up with new mechanisms. Now, China can do first-in-human or clinical trials so much faster and with so much flexibility, that its actually giving the US FDA a lot of pressure. This topic was brought up so many times in 2026. Albert Bourla, Pfizer CEO, talked about it, and the former commissioner of FDA, Scott Gottlieb talked about it, publically criticising the FDA: “you are too slow, you are too bureaucratic, you are slowing down our biotech industry.”
China has this unique system called the investigator-initiated trial (IIT). If you want to do, let’s say, AAV-based (adenovirus-associated) gene therapy for some rare diseases in the US, good luck! It’s very difficult. The FDA is going to require you to have a CMC package (Chemistry, Manufacturing, and Controls), prove you already have enough supply, you have a clinical GMP (Good Manufacturing Practice) facility, you have enough safety data, and you have protocol which costs tens of millions of dollars – all of this takes a long time.
The investors and the biotechs are really miserable right now if they are in the gene therapy space in the United States. But in China, it’s very different story. China has this IIT (investigator-initiated trial) system really designed for cell and gene therapy, without having to have a full blown commercial GMP facility, without even needing approval from the NMPA regulators.
If you have really encouraging data pre-clinically, and if you can really prove that it’s likely going to be very safe, with tremendous clinical benefit, then the hospital IRB (institutional review board) will green-light you, and you can get the trial done in a local hospital quickly.
This has already happened: EsoBiotec, the Belgian company, were doing in vivo CAR-T for a lentiviral based candidate. They did their first in human trial in China, and after just a few patient data, it looked really fantastic. AstraZeneca put down up to $1 billion to acquire the company, and it became the first clinical stage in vivo CAR-T based on lentiviral vector in the world. As of today, big pharma has done five acquisitions of the in vivo CAR-T company. Based on public information and private information, I can tell you that the majority of them have already started or will start first-in-human clinical trials in China.
Related:
5. Deal pricing
Ruby: You were dropping these very high value deal numbers, with a lot of money on the table. When you’re looking at this landscape, have you thought: this is underpriced, or they paid too much there? Is there a big difference between some of these deals in terms of how realistic they are?
Leon: As a deal maker, I always think that the price is fair in that specific situation. The big pharma challenge is real, there’s a hundred billion dollars of patent cliff. The induced revenue loss is real. We all knew that Sanofi’s CEO Paul Hudson was on the way out. Every analyst will tell you the problem is, despite the revenue in recent years looking pretty decent for Sanofi, their patent-cliff problem was not being solved. That’s probably one of the major reasons he was asked to pass the baton to someone else So that’s a structural challenge for big pharma. They have to buy stuff, in-license new stuff, to maintain their pipeline and maintain their future revenue.
With that being said, before, China biotechs were honestly not very savvy in terms of doing deals with Western big pharma or biotech. This why some people called it arbitrage opportunities, and pretty much we call it the “China discount”. But in 2026, all of a sudden, the China discount - at least look at those leading deals out of China - is totally gone. And instead of getting China discount, now you have to pay the “China premium”. It’s crazy! They actually compared the recent transactions, especially asset transactions of license deals with Chinese biotechs compared to other BD deals of the same period of 2026 with Western companies of the same stage, say phase one versus phase one, phase two versus phase two - then you pay more to get a Chinese asset.
That’s a very interesting phenomenon, but of course, it is a little bit of FOMO. We know that a lot of big pharma right now have full-time employees of their BD team in China. One of the largest European pharmas has a BD team of 10 people based in China, go through all those Chinese assets, even talking to boutique Chinese companies. So as a deal maker, I always think: how can you get a good price if I have multiple bidders? It doesn’t matter if it’s Chinese asset, European asset, Indian asset or Singaporean asset, as long as you have, let’s say two or three bidders, you can use them against each other. You say that Party A gave me 100 million dollars, you have to match as Party B if you want to win. Pay more, it’s very straightforward. So because you have so many buyers in China chasing after the same hot assets, of course the price is gonna go up.
Like I mentioned, the Chinese Healthcare Investment Conference, there are so many investors also chasing after early-stage Chinese assets, because typically big pharma don’t feel like taking such high risks. It’s going to be the investors doing that, but now, investors have to compete against each other for the same early stage assets, even if higher risk. So of course, early stage asset price is going to go up as well. It’s just competition in the marketplace. expected market dynamics.
Ruby: So moving from China discount to maybe China premium, as you said, in some cases. there is all of this hype and an urgency to get hold of the right assets. But then at the same time, all of that perhaps is still sector specific. And in the general broader audience landscape, at least in media narratives and non-biotech specialist spaces, we’re still trying to convince everyone that Chinese biotech is even good enough quality or viable. So this widening gap between reality and the sector versus the general audience, I guess, that will just change with time as we see pipelines mature and patients actually taking these China origin medications, having their lives changed for the better around the world, then general audiences will start to see the evidence. And also, then, we’ll start to see the risks and the challenges come up as the reality lands. Do you find yourself a lot of the time still jumping between these two widening worlds?
Leon: Honestly, the generalist is really not a serious player in biotech space. Even in the US, if you look at who is really driving the biotech industry, it’s really the specialists. The specialists have so much capital, they don’t necessarily need a generalist. Generalists are placed mainly in big pharma, if you ask those generalists in government fund, sovereign fund, or retirement fund, they manage a hundred billion dollars. They want to invest their minimal check size of $200 million. How many biotechs can take that? They can’t. So what general investors would do instead is to look to big pharma. That’s it. That’s what a generous investor, especially a public investor is really doing.
Biotech is really driven by specialists. Now it’s even more clear. So I don’t really worry about retail investor in today’s situation .The science of biotech already complicated enough, and now you’re throwing in in China and geopolitical issues in it too. Honestly, I don’t recommend any generalist sitting in the US who doesn’t know any Chinese, to invest this sector. It’s too difficult!
6. Deal-making drivers: buy side and sell side
Ruby: If we talk a bit more about your role as a dealmaker and in supporting investors on both sides of the world, can you take us behind the scenes - on the drivers, whether that’s on the buyer side, biopharma, big MNCs and top tier biotechs, what is changing in terms of how they search or screen? you mentioned marriage, decades long partnerships, slow building. But now that everyone’s getting conviction on China origin, China linked assets, what is changing?
And on the investor side, you’ve just mentioned maybe not the general investors, in your opinion, but at least the specialist investors. the funds are deciding whether the China risk, China exposed assets, even if they’re high risk, whether that’s investable next five years, and what they’re under or overwriting in terms of the science, the execution, geopolitics, anything else.
Leon: First of all, the big pharma, we call them strategic, including profitable mid to large cap pharma and biotech in the world. So for them, they typically have a capability to do multi-billion acquisition either as a company, which I saw this morning, Servier, the second largest pharma company from France, acquired the US based company, Day One.
I put that into this category, where they are pretty adventurous, have been China for a long time, and fully embrace China from A to Z. They even talk to the universities. One of the big pharmas, their China trip organiser asked me to connect them to Westlake University, which is now one of the best universities in China. They wanted to talk to the professor. It was fascinating how deep they are in Chinese biotech universe – not even just Chinese biotech, but Chinese academia - they even know that Westlake is good at the biological basic research. Very surprising how the front-running pioneers, even big pharma, really know China.
But of course in another category, they are a little bit more conservative, they are still worried. The most they want to do is just a license deal. So we see that wide spectrum: there are early movers, there are late adopters.
For investors, it’s very similar. There are some investors, they were created 15, 20 years ago when there was a first wave in the belief that China biotech might be promising. The best example probably is Lilly Asia ventures, created with the big help of Eli Lilly. It was a semi-corporate venture to begin with, and later on became an independent private investment firm. They’re one of the most influential biotech investors in China.
So this is a China dominant one. There are also some US ones. Probably the number one most active investor from the US is Orbimed. They never left China even when other healthcare tier one investment firms did, due to geopolitical, COVID, whatever reasons, the others left. But Orbimed never left, so now they have a huge advantage when China biotech is picking up. They got in a lot of good companies early on in. And of course, there are other tier one funds, who never thought China was important. So now they are playing catch up. And a lot of funds are actually opening their incubators in Shanghai or Suzhou. Some of them are shutting down their incubators in the Bay Area or in Boston. It’s happening because again, no brainer. If you are a capitalist, you just look at ROI. With $10 million in Kendall Square in Boston, you will not even have cell line development (for your biologics). But in China, with $10 million, you can probably already dose first patients in IIT trials.
So as an investor, if I get some technology, say from Harvard, I can either do the &D development in Kendall Square, so the professor can visit, or I can do it in Shanghai or in Suzhou, where the professor can still visit virtually, via ZOOM call, not a big deal. This week, actually, we saw the reverse emerge, of Candid therapeutics with Rallybio. That’s how they’re to go public. The article that described this IPO through a reverse merger, it called Candid biotech a “San Diego- Shanghai biotech”. This definition is very interesting. So Candid is one of the superstar private companies, and they are falling into this big category called “buy and build” biotech. Many companies in this category in the US start with a pipeline, they license the pipeline from Big Pharma.
But in Candid therapeutics, their four pipeline assets were licensed from China. Yet different from most other US biotech, they’re not a classic NewCo story (a newly formed company built around a licensed or spun-out asset). Most NewCo in the US, once they license the assets, they start developing in the US or in Europe.
But Candid decided: no. Since China is so efficient with early stage clinical development, why not just develop those assets in China? So they built a team from scratch in Shanghai, and they have multiple trials running in China. The company went out of a stealth mode in late 2024. Now they are going to IPO. How fast is that? The valuation is crazy. They start with $370 million series A. And before they reverse merge IPO, they raised another more than $500 million. So they raised $900 million, almost $1 billion. Now you can do the math of what their valuation going to be. Very interesting. And if you look at their investors, these are all the best investors, most of the best investor from the US, from China, Europe. So it’s big party, the most popular party, all those investors want to pile in.
7. From the perspective of Chinese biotechs
Ruby: So we’ve talked about the biotechs on the Western side and the investors, but then also thinking about the Chinese assets and the companies that are producing these. As they’re now becoming increasingly selective about who they partner with, what are you seeing from their point of view as they’re gaining experience, becoming choosy in who they think has a good reputation to let them land better outside of China?
Leon: It’s about option. You can pick and choose. Right now, especially if you are Innovent, if you are Hengrui, or if you are CSPC, all those hot deals have multiple suiters, multiple bidders. Don’t think that the Chinese don’t know how to do deals anymore. They really know how to do deal, because their deal makers are trained in the US or in Europe, They know how the trade secrets of doing deals can be easily transferred just by building a team. Just get the talent to come over, and they’re going to streamline everything, and you can have globally competitive deals. A lot of investment bank and the top transaction lawyers are helping those China biotech biopharma as well. The talent is so fluid, in the end it is really about whether your asset is globally competitive, and can really fit the big pharma pipeline. China biopharma now have zero knowledge gap.
But again, the caveats. People say that China right now has somewhere between 5,000 to 8,000 pharmaceutical focused companies, including big pharma and biotech. That’s a huge number. But if you look at the companies who really have done meaningful deals in the past three years, the numbers are less than 200. So the percentage of really good China pharmaceutical or biotech companies is really, really low. Let’s say you talk to 100 Chinese biotech companies, the majority of them will be not so good. If you are not very selective.
Ruby: That’s a big difference proportionally. For a Chinese biotech company to choose to partner with the right Western multinational, is it that perhaps that multinational has already got expertise and market access in that particular type of pipeline? Or is it because they’ve got a longstanding relationship built on trust? Or is it that the size of the deal is the best? Or a combination of those and more. Hard to say?
Leon: This combination - all of this and more. It’s really case by case. Not because the company just said, believe my asset is best, I can sell it. But if you plan really well, sometimes you can envision what the future is going to be. And then if all the stars line up, by the time you are ready to transact and your asset becomes really hot, then that’s where you can do pick and choose. Then the decision process criteria, who they’re going to work with, is really everything you mentioned and more.
So it’s really hard to say, but the bottom line is the company who made the right decision two or three years ago to start a project. Right now, let’s say they get into phase one, you’re one of the hottest of the field. They’re have a really strong upper hand. There definitely is a seller’s market in that case. That’s where those big deals really come from.
Ruby: And then in terms of your job, delivering these deals and making those connections, as we’re saying, there’s not only excitement and realisation, but also hype. Is your job becoming easier now that you’re doing less convincing and more practical doing? Or is it trying to sort through the greater amount of things on the table?
Leon: I have two mandates. I have a buy side mandate, which is Western biotech companies and investment funds. I have a sell-side, when I represent my sell-side mandate, which typically is a China biotech company, phase one ready or phase one, phase two stage asset, we try to find a Western partner, outlicense our China rights. In that case, the good news is that I don’t need to tell them anymore that Chinese biotech research and assets are good. That conversation is now totally skipped, I don’t need to that anymore, they already are China believers. I barely talk to China skeptics.
A typical BD conversation, you tell them why this is a good fit for your programme, why this is going to have tremendous value if we work together because we are small, or we cannot do early stage clinical development, we cannot do commercialisation, but you guys can do that. We create a value together, That’s the typical stuff. The difference between representing a US biotech or European biotech to out-license to big pharma, and doing this for China biotech, is really small now. So that’s the sales side.
On the buy side, I represent the small guys, to buy Chinese assets. So actually our job becomes harder and harder, because the big guys, the strategists, are flooding China with their big cheques. We’re always joking that in the building process, any time big pharma joins, we leave. We get priced out. We get gentrified by the big pharma. They do the terms sheet of upfront pay much bigger than ours. So that’s always a little bit challenging.
However, the market has also become stabilised. Big pharma have all the cheques, but their risk tolerance is much lower than us, than my clients. My client typically their job is taking risks. Our chequebook is smaller, but our stomach for risk is much bigger. So that’s where we play.
I think that before, people in general labeled all the China biotechs as having less negotiation power, but now that label pretty much is gone. Again, a lot of China biotechs, if you legally look at them, they are not really Chinese companies at all. They’re either Cayman Island registered companies; some of them are just straightforward Delaware companies with operations in China; or the IP resides in the US or Europe from the beginning. So legally speaking, actually a lot of so-called China biotechs are European or US biotechs with R &D operations where the “R” is in China.
Even if you really define big pharma based on where they do R &D, AstraZeneca has five R &D centres, one in Europe, two in US, and two in China. So how would you call AstraZeneca? Is this a Chinese company, an American company, or a British or Sweden company? What’s your definition? They’re listed (as a public company) both in London and also in New York.
Ruby: As everyone is doing cross-border and having multiple locations around the world, it seems that strategically you call yourself whatever region is optimal for the situation you’re in. Whether that’s geopolitics, capital, whatever.
Leon: R &D is intrinsically and by design global. And somehow the politicians, the government need to pigeonhole them into different country, which just does not reflect the nature of this business.
Ruby: Yes, that’s such a great point. Health, biotech, innovation has always been global. Institutions and geopolitics, segments it all in challenging ways.
8. Europe vs US
Ruby: Just picking up on European versus American investors, regional differences in appetite or perception. Can you share a bit more about that from what you’ve seen?
Leon: I would say one or two years ago, there was a misconception, especially by some American investors, or even especially American politicians, who believe that the US is the dominant buyer of Chinese biotech asset innovation. They said, once we cut off the US-China license deal, China is about to die.
Everyone in our industry was laughing so hard, because we know the numbers. Actually, in 2024, the Europeans spent more money than Americans in licensing Chinese assets. 2025, some people argued that because of the potential US ban of Chinese assets going to the US company. there’s a front load of Chinese assets in case this happened. And you remember last year, the draft executive order from Trump administration to ban Chinese license deals, someone saw that, and they front loaded. So last year, I think the US probably spent slightly more money on Chinese assets than Europe.
The China biotechs, they have option, So a good example is the largest China outbound deal by value - it’s not with the American companies, it’s with AstraZeneca. AstraZeneca and CSPC signed the largest deal ever with China Biotech, $18.5 billion. And also remember, when did AstraZeneca sign the deal? when Minister Starmer visited China. So that was one of these diplomatic icings on the whole thing.
So we cannot say to Chinese biotechs, we demand you do this. They would just say, if you’re really so demanding, I won’t do business with you, I will do business with Europeans. That’s why people realise banning the US-China dealmaking would be such a bad idea for the US biotech and biopharma and US investors. The net is a huge downside for the US industry. So we should just figure out a way how to live with China. Take something they’re very good at, take something we are really good at, and let’s just work together, create value together for patients around the world. I think that’s where the mentality should be.
And you can also tell right now, instead of blaming China: “they steal, they cheat, they subsidise”, which is just a little bit true, most of it not true, right? Now there’s sharp contrast between how China does things and the US does things. The US gas realised our FDA is the problem. I mean, the Chinese have IIT, we have nothing like that in terms of equivalent pathways.
For a US biotech company, if they are invested by some well-informed investor, they will tell them to do the first in human trial outside of the United States, and to recruit patients as much as possible outside of the United States until trials must be done in the United States to satisfy regulations. It used to be Australia as a preferred choice, but now China gradually become the preferred choice because of patient population, because of cost, because of everything.
Ruby: From the China side, what do they see as an advantage of Europe versus America? Or are there not really, because as we just said, every company is actually global?
Leon: So I think in the US, our position makes it very clear that biotech is a bargaining chip. So whenever President Trump and President Xi need to negotiate, this is on the table. It’s just the risk. Biotech needs certainty. And we are in such a high-risk industry. Any regulatory geopolitical uncertainty can destroy a company, period, and that’s exactly what happened, domestically in the US. That’s why a lot of investors say they will never stop investing in cell and gene therapy in the US, just because of FDA, unless they’re weak.
So the same thing for some, not all China biotech biopharma. If they want to have five year, ten year collaborations with a partner, they want to make sure the partner is going to be located in a stable environment. In that case, Europe probably has more attractive potential than the US. There are a few straightforward European acquisitions of Chinese located or based biotech company in the past couple of years: Gracell was acquired by AstraZeneca, ProfoundBio by Genmab, Biotheus by BioNTech, and another was SanReno, acquired by Novartis. All those buyers came from Europe. That could be just an accident… you can form your own opinion!
Related:
9. Execution
Ruby: And then moving on to the execution, after the deal and the partnership gets going, what signals can we use to predict the potential of realisation or the risks of these cross border partnerships, for Chinese biotech with enough capital, enough expertise, if they decide to utilise their own manufacturing and supply chains on the ground in the US market, rather than partner with a Western MNC to access patients and providers? Obviously, maybe it’s early on for some of the deals we’re seeing now, but the ones that have been going for a couple of years now. What are you seeing being indicators of them being successful or less so,
Leon: If you really want to become commercial stage company for the Western market, even if the innovation originates from China, you have to manufacture their stuff preferably in the US or Europe or in allied friendly countries. So that’s one of biggest barriers for the China biopharma biotech to climb the value chain, and to become a late stage or commercial stage for the Western market.
BeOne is now a Swiss company, it’s not a China company legally. So they totally uprooted themselves and put themselves in the Western ecosystem. So I don’t treat B1 as a China biotech at all, but a European or global, mid-sized biotech. so all the baggage other China biopharma have, they don’t. But for the others, like Hengrui, they do have the challenge. Their manufacturing, their R&D efficiency, cost advantages and so on, only exist if they do that in China. If they leave China, they leave their core competitive advantage, that’s why they have to partner with the Western biopharma.
So that’s why I don’t worry much about the late stage biotech or strategic big pharma, big biotech from the West. They will not have much pressure from the rise of China biotech, because actually, especially between US and China, the geopolitical issues create a pretty insurmountable barrier for China biotech to really climb the value chain. To use the jargon of NPV (net present value), the majority is going to stay with the US biopharma, with large biotech, just because they control their manufacturing, they control their commercialisation and late stage clinical development capability.
10. Looking ahead
Ruby: And finally, the forward view. We’re in March now and we’re looking ahead at the rest of 2026 and beyond. From today’s vantage point, what are you watching for, in terms of the momentum going forward?
Leon: The biggest one is really still the regulation and geopolitical risk factors. We are a global business, and anything that disrupts the global collaboration and money or capital flow or change of knowledge is going to be pretty substantial for our industry. So that’s definitely number one risk for China-to-West collaboration.
But of course, people get into biotech because there’s a lot of upsides in helping patients around the world. I would still closely watch the deal flows, either licensing deal or R&D collaborations like what Eli Lilly did with the Innovent. If those kinds of deal flows continue without any sign of slowing down, the truth is that the American corporate executives are so well informed, and they have a direct line to the White House, to all those Congress, policymakers, lawmakers, and they make informed risk taking. If that continues, probably their evaluation of risk exists - but it’s not that big, which is good.
I will also look at the exchange of talents from the West and China. whether they continue, whether collaborations also continue. If that’s the case, I think that misconceptions and misunderstandings will be less likely to happen, or less likely to mature to such a stage that some dramatic detrimental decision would be made from either side of government or policymakers. So those are three things that I’m most curious about.
Watch or listen here on substack or Youtube, and/or subscribe on Spotify & Apple Podcasts.